PFMD was joined by new members in 2019 including Astellas, HRA, Eli Lilly, and Gilead.
Patient Focused Medicines Development A global collaborative non-competitive coalition to make patient engagement the new normal
Introduction from the Programme Director
Breathtaking, collaborative, engaging and progress are some of the key words to sum up 2019, though, they too fall short to describe the excitement of the growing global determination and efforts invested in systematic patient engagement and making it happen with all stakeholders, throughout the health landscape. Moving from thematic frameworks to the nuts and bolts of doing patient engagement is the theme of the new decade as we move on to 2020 and PFMD is at the forefront to collaboratively lead and drive this change with co-created and dedicated tools and training to enhance implementation and integration of systematic, meaningful and effective patient engagement.
It is essential that we as a community focus on collaboration and coordinated efforts, build on synergies that will help us reach our shared purpose in improving global health goals through increased engagement. This is one of PFMD’s main operating principles and now more than ever, it will be our responsibility to connect the dots and fragmented work for the global good.
From geographic expansion to extending patient engagement into devices, digital health, big data and coordinated regulatory and HTA efforts - 2020 brings exciting new opportunities to PFMD.
2019 Achievements
Membership
Multi-stakeholder Co-creation Working Groups initiated and ongoing
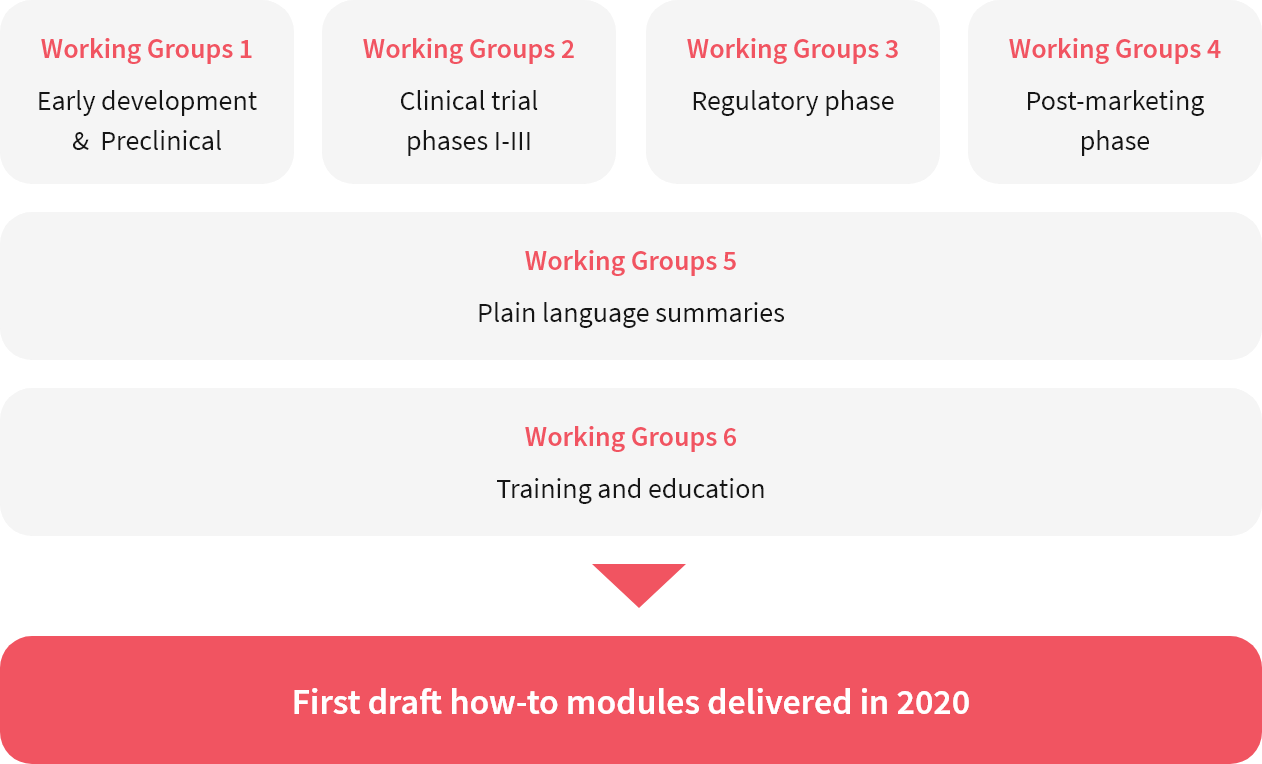
PFMD Multi-stakeholder Co-creation Working Groups were first convened in February 2019 to deliver how-to guides for specific activities in the medicines research and development continuum. These six Working Groups, which utilise the 7 Quality Criteria (from Patient Engagement Quality Guidance) each focus either on a specific medicines development phase or on a transversal activity that exists throughout the continuum. The outcomes expected during 2020 will be very practical guides for stakeholders to engage patients in these activities.
By the end of 2019, 82 contributors from 49 organisations are part of the PFMD co-creation Working Groups.
Successful launch of the patient engagement training
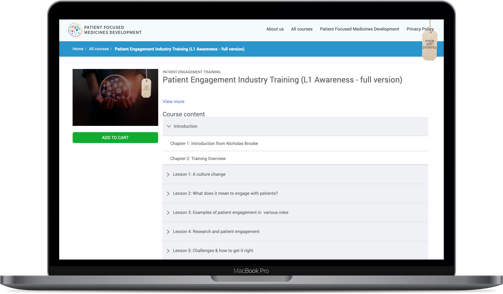
This first patient engagement training module was launched in September 2019 and has been developed out of a need for a co-created patient engagement awareness & training programme for industry professionals, matching both user & company needs.
The training is for anyone in a pharmaceutical company to understand patient engagement, its value and their role in making it happen.
Co-designed with 13+ organisations, it can be used as an awareness tool for culture change for reaching the early and late majority within the pharma industry.
By the end of 2019, we had a total of 1338 users on the platform with 994 training completions.
Synapse
For understanding the patient engagement landscape, finding people, resources, organizations, events, and initiatives, Synapse continues growing into a complete and comprehensive patient engagement mapping and networking tool.
An inter-connected ecosystem with close to 1,700 people, over 400 organisations, more than 370 initiatives and 350 resources in 2019. The tool also enables its users to monitor and manage their organisation’s patient engagement activities and relationships within a private section.
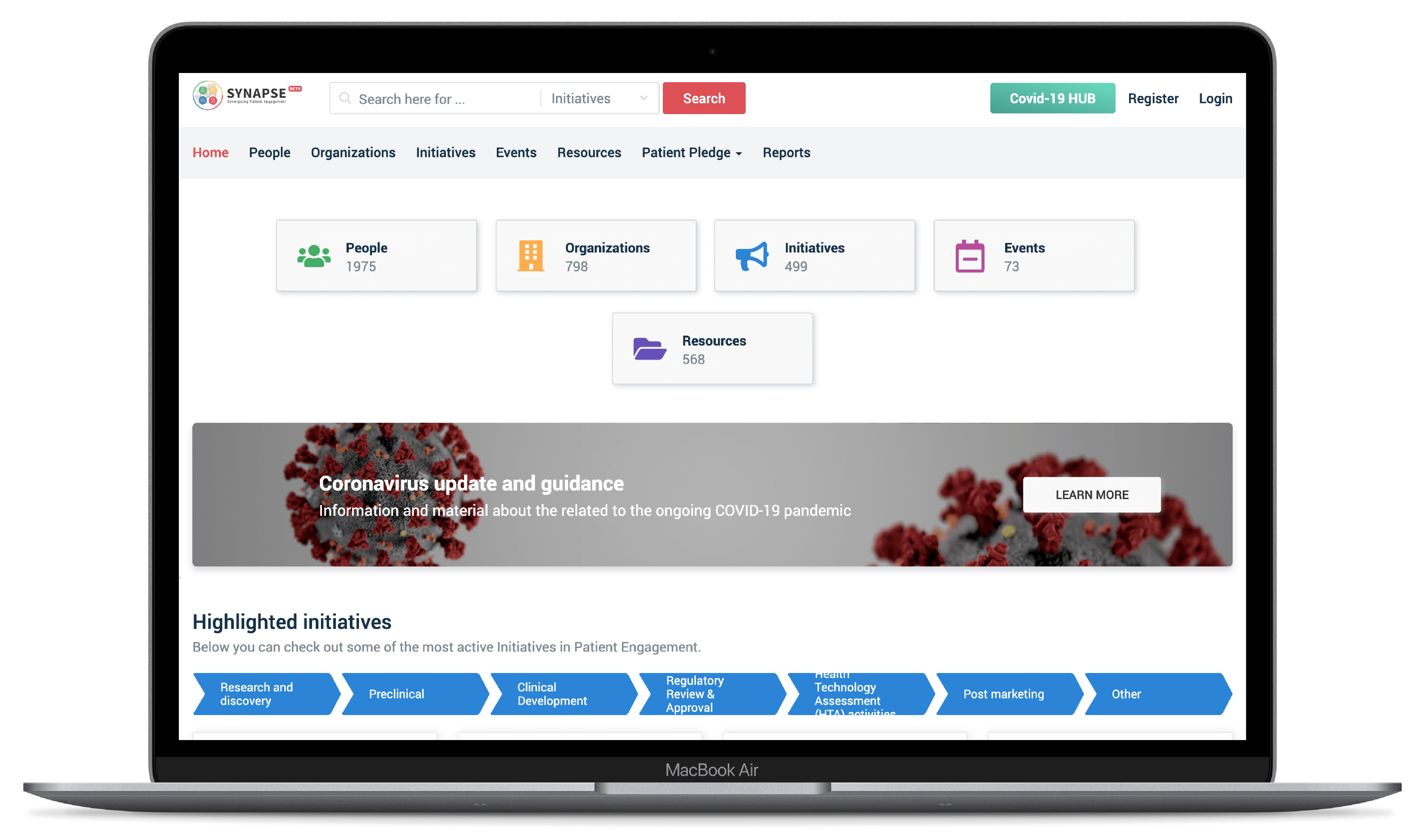
- Continuous usability improvements
- PE Quality Guidance integrated in Synapse initiative questionnaire
- Improved organisation management: categorise users in business units
- Conditions pages (beta) improved with integrated repository for clinical trials and scientific publications
- Improvement of global reports started
- Infrastructure upgrade for platform scalability
- Events calendar (beta) released
- Improvement of global snapshot reports
- Integration with PE Learning system begins
- Book of Good Practice second edition released in Synapse
- KPI dashboard
- Partnership improvements begin
- Condition pages improvements begin
Communication impact
To continue impactful communications to increase awareness of systematic patient engagement and available tools, PFMD keeps on expanding its digital outreach and relevance with strong ongoing engagement
The Patient Engagement Open Forum from PFMD point of view
The Patient Engagement Open Forum welcomed patient engagement experts and enthusiasts in a two day event where we explored patient engagement beyond aspirations and worked in a multi-stakeholder context to make it happen.
The event was co-organised by PARADIGM, PFMD and EUPATI.
sessions
including topics like:
- Best Practices in Patient Engagement,
- Communication & Capacity Building,
- Legal Contracts,
- Fair Market Value,
- Patient Engagement Framework and
- Working Groups for “How to” Modules
#PatientEngagementTrended in Beligium for two days (18-19 september)
PFMD Social media impact
200+ Delegates
Including Patients, Patient advocates, Pharma industry, MedTech industry, academia, HTA, Researchers, Regulators, Trade, Healthcare professionals, Media
Book of Good Practices
The second edition of our popular Book of Good Practices was released on November 4, 2019, and included 5 new initiatives.
Find more here: Synapse

Publications

If you are curious to find out how the initiatives for the Book of Good Practices (BoGP) were selected, check out our publication in DIA Global “Patient Focused Medicines Development: The Book of Good Practices Evolution of Methodology”
The Patient Engagement Quality Guidance and the Book of Good Practices are practical resources which will be continually updated in response to user feedback. They are not prescriptive, but rather based on core principles, which can be applied according to the unique needs of each interaction and initiative. For in-depth discussion, check out our publication “Co-creation of patient engagement quality guidance for medicines development: an international multi-stakeholder initiative” published in BMJ Innovations with 2,821 abstract downloads.
Our 2020 Approach
As we step into the third decade of the 21st century, PFMD’s projects will continue to engage a great number of stakeholders in co-creation and collaboration efforts. The New Year brings many new beginnings, one of which is the webinar series designed to enhance community engagement through knowledge sharing.
The ongoing workstreams are expected to deliver outputs throughout the year starting from the very first months.
PFMD Multi-stakeholder Working Groups: PFMD multistakeholder Working Groups are co-creating “how-to” guides for specific activities in the different medicines research, development and delivery phases. The outcomes that are expected to be released throughout the fall of 2020 will address a wide range of practical Patient Engagement topics.
The Book of Good Practices (BoGP) - Third edition: The BoGP will be collecting good patient engagement practices for the third edition projected to be released at the end of 2020.
Patient Engagement Training for Industry Professionals: The short version of Level 1 will be released in 2020, while 2020 will also see the launch of training level 2, which will be a very practical and interactive training module on how to operationalise patient engagement, offering hands-on training with practical tools, tips and tricks.
The development of training level 3 will also begin in 2020; this is more of a catalogue of advanced and specific mini-modules (marketplace) including several topics. These topics will be confirmed based on needs’ assessment and will take place in collaboration with different partners.
Fair Market Value Calculator: 2020 will see the launch of the Fair Market Value (FMV) calculator by the National Health Council (NHC). The FMV calculator will be designed to help determine the compensation of patients (and caregivers/family members) and patient groups involved in ‘patient engagement activities’ taking place between patient organisations and/or unaffiliated patients and private companies.
PFMD will also plan for an adapted calculator for European use, in collaboration with European Federation of Pharmaceutical Industries and Associations (EFPIA) and Workgroup of European Cancer Patient Advocacy Networks (WECAN) based on the United States Fair Market Value calculator.
Country cards: PFMD will convene a taskforce with patient engagement experts, compliance and legal experts and commence work on summarising national patient engagement rules and tools.
Collaboration with Patient View: We will develop a co-branded PFMD-Patient View survey on Patient Engagement Quality Guidance, which will be globally accessible in 19 languages and targeted at 40.000 patient organisations. The outcomes can be used by corporate organisations as a tool to measure and plan for patient engagement.
Synapse improvements: The Synapse networking and mapping tool will continue evolving to match user needs. This will include updates in initiatives and reports,access to pages, improved organisation networks, and more.
Stakeholder engagement and collaborations: Co-organisation of the Patient Engagement Open Forum with IMI PARADIGM and European Patients' Academy (EUPATI). A collaborative effort to connect the patient engagement ecosystem, share good practices, drive for more systematic patient engagement and defragment the landscape to promote collaboration across stakeholders and initiatives.
Collaboration with IMI PARADIGM to deliver a legal toolkit that aims to take the Guiding Principles and the four Reference Agreements to the next level by providing plain language explanations for each clause. This toolkit aims to help patients in navigating the legal landscape when collaborating with the pharmaceutical industry.
Co-creation of the Fair Market Value Calculator to systematically determine a fair compensation for patient input with the National Health Council (NHC). The preliminary excel calculator is in review with NHC and will be further reviewed by the steering committee for beta testing.
Co-development of the Guiding Principles booklet and Advisory Board Reference Agreement with the Workgroup of European Cancer Patient Advocacy Networks (WECAN), Myeloma Patients Europe (MPE) and over 10 pharmaceutical companies for the ‘Reasonable agreements for partnerships between patients and pharmaceutical companies’ project.
The PFMD Board
The PFMD Board Members consists of:
-

Nicola Bedlington Secretary General, European Patients Forum -

Veronica Todaro Vice President, National Programs, Parkinson’s Disease Foundation -

Graeme Johnston Patient Advocate, Individual/ Patient -

Alexandra Moutet Global Head of Patient Affairs, UCB -

Roslyn Schneider Global Patient Affairs Lead, Pfizer -

Andrew Benzie Head of Patients in Partnership Programme, GSK -

Katherine Capperella Global Patient Engagement Leader, Janssen -

Rebecca Vermeulen Head Global Medical Liaisons and Communications, Roche -

Elisa Ferrer Patient Engagement Manager, EURORDIS -

Kelli Collins Vice President Patient Engagement, National Kidney Foundation -

Lode Dewulf Chief Patient Officer At Servier Group -

Paula Wray Senior Public Involvement Manager, NIHR INVOLVE -

Janice Nissen VP, US Regional Customer Operations, Vaccines At Merck -

Cindy Grossman Director, Science Of Patient Input, FasterCures -

Begoña Nafría Escalera Patient Engagement In Research Coordinator- Hospital Sant Joan De Déu -

Laura McKeaveney Global Head Of Patient Advocacy At Novartis
The PFMD Executive Committee Members consists of:
-

Nicholas Brooke Executive Director -

Marc Boutin Chief Executive Officer, National Health Council -

Tony Hoos Head Of Medical, Europe, AMGEN -

Jan Geissler Patient Advocate, EUPATI
The PFMD Team
The PFMD Team consists of:
-

Helena Harnik Operational Director -

Jean-Christophe Capelle Financial Director -

Chi Pakarinen Project Manager -

Eric Blanchet Operations Director -

Roxana Radu Communication Manager -

Lise Brooke Growth Hacker -

Gulwish Ahmed Communications Coordinator -

Laila Deeb Web Developer -

Anne-Marie Hamoir Senior Consultant -

Ify Sargeant Scientific Editor -

Danielle Derijcke Program Manager -

Bernadette Hendrickx Scientific Advisor -

Bonaventure Ikediashi Project Coordinator -

Jeremy Jamar IT Architect -

Daniela Luzuriaga Ubilla Project Coordinator

















































